Case Report Valver
Balloon Aortic Valvuloplasty and Tavi Post-Dilatation with Valver Balloon Catheter in Adult with Severe Aortic Stenosis in Transcatheter Aortic Valve Implantation.
Baseline
A Case Report on Transcatheter Aortic Valve Implantation in an 80-Year-Old Patient with severe aortic stenosis. The mean left ventricular ejection fraction was 54%, the mean aortic valve pressure gradient (PG) was 75 mmHg and the mean AVA was 0.65 cm2. The TAVR procedure was performed in interventional cardiology at Clinica San Rocco in Brescia city, by dr. Emiliano Boldi. TAVR endoprosthesis, Medtronic CoreValve™ Evolut™ R (Medtronic, Minneapolis, MN, USA) Balloon aortic valvuloplasty, Valver® balloon catheter (Balton, Warsaw, Poland).
Interventional procedure
To assess the real expansion of the TAVR endoprosthesis, 2 Valver® balloon catheters were used,the first one is Valver® 20x40 mm for Aortic Valvuloplasty (Fig. 1) and the second one is Valver® 23x40 mm for endoprosthesis Post-dilatation (Fig. 3).
The procedure was performed from the femoral artery access under local anesthesia. The balloon diameter choice was left at the operator’s discretion; however, its nominal diameter did not exceed of the aortic annulus diameter measured in TC images. Normally, the nominal diameter of the valvuloplasty balloon diameter (Pre-dilatation) is chosen according to the minimum diameter of annulus, while the nominal diameter of the post-dilatation balloon is chosen according to the average diameter of the annulus. Routinely, the balloon is inflated with a manual syringe and the aortic balloon inflation is performed most frequently during the rapid pacing of the right ventricle, usually at 160 to 180 bpm.
The pre-dilatation is not a fundamental requirement in TAVR, but the physician has decided to predilate for three reasons. the first reason is due to the high mean pressure gradient (75 mmHg).
In this case is preferable to predilate. The second reason is due to the small valve area (0.65 cm2). In this case, the valvuloplasty helps gain more valve area and less compression of the prosthesis after deployment. The third and final reason is due to the uniform dispersion of calcification on the aortic valve.
Device description
Valver balloon catheters (Balton, Warsaw, Poland) have been certified with CE mark and have gained a marketing authorization valid throughout the European Union since 2008. They are available in different sizes (a diameter of balloons ranges from 5 to 35 mm and length from 20 to 60 mm) and are compatible with 0,018-0,038” guidewires and 4-16 Fr introducer sheaths.
They are low profile and low-pressure balloons (for example, nominal balloon diameter 8 mm/ length 2 cm needs shaft diameter 4 F, guidewire 0,018”, its nominal pressure is 2 atm, RBP 5 atm. Generally, according to producer information a nominal pressure ranges from 2 to 3 atm and a burst pressure from 3 to 5 atm.
DISCUSSION
This case showed that the BAV procedure with Valver balloon catheters is a feasible
and safe option.
The use of balloon aortic valvuloplasty (BAV) has increased with the development of transcatheter aortic valve implantation (TAVI) to medically optimize patients prior to procedure. Use of a low profile compliant valvuloplasty balloon has been demonstrated to have adequate BAV results with smaller sheath sizes.
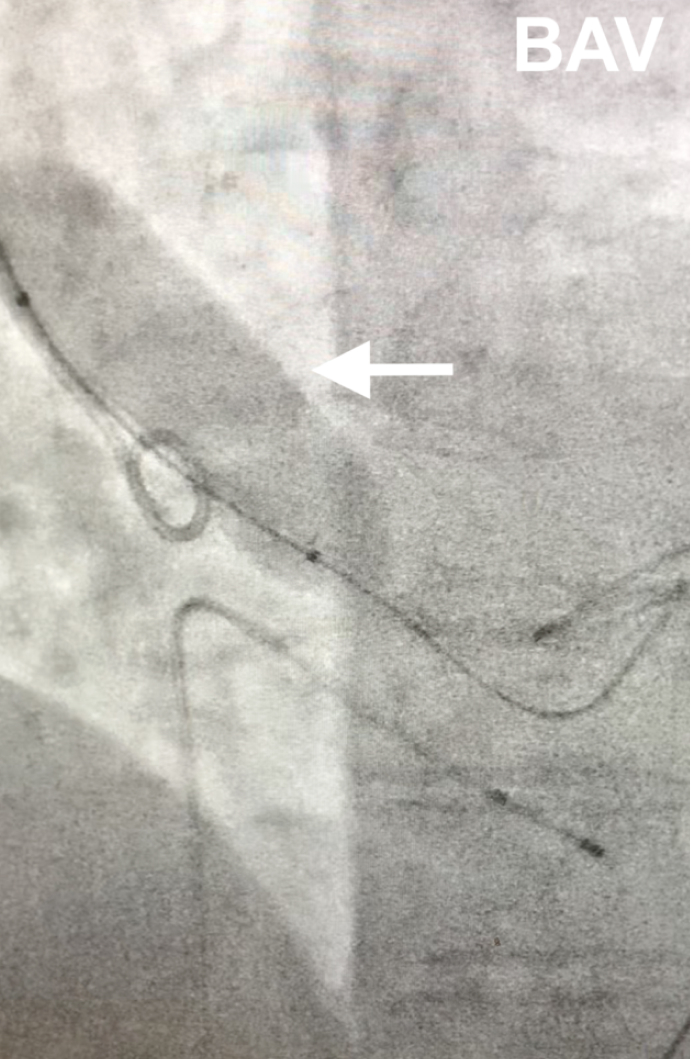
Fig. 1- Balloon aortic valvuloplasty under fluoroscopy using Valver® balloon catheter 20x40 mm (Balton, Warsaw, Poland)
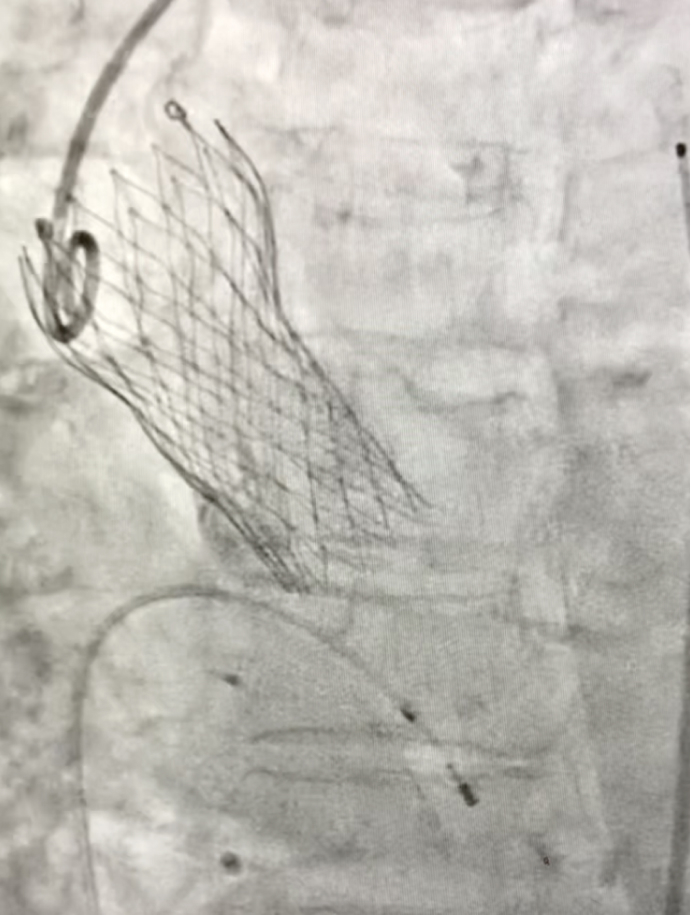
Fig. 2- Under-expanded CoreValve™ Evolut™ R 26 mm aortic valve (Medtronic, Minneapolis, MN, USA)
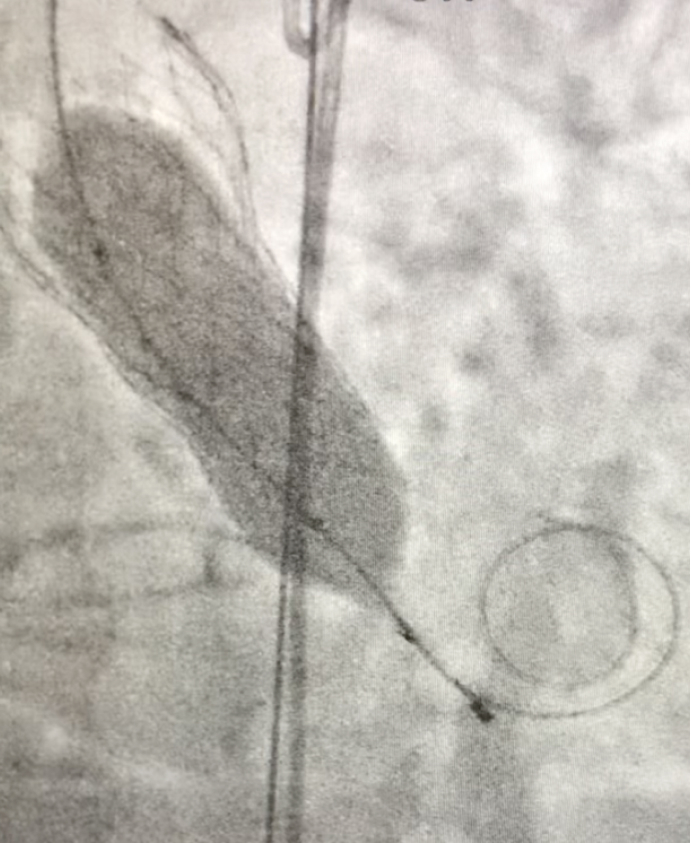
Fig. 3- Balloon aortic Post dilatation under fluoroscopy using Valver® balloon catheter 23x40 mm (Balton, Warsaw, Poland)